result



Trial Run

Precision-engineered therapies take their first steps in early-phase clinical trials at the Cockrell Center for Advanced Therapeutics —translating science into hope for patients in need
GIFT USHERS IN CLINICAL TRIAL GROWTH WITH A HUMAN TOUCH
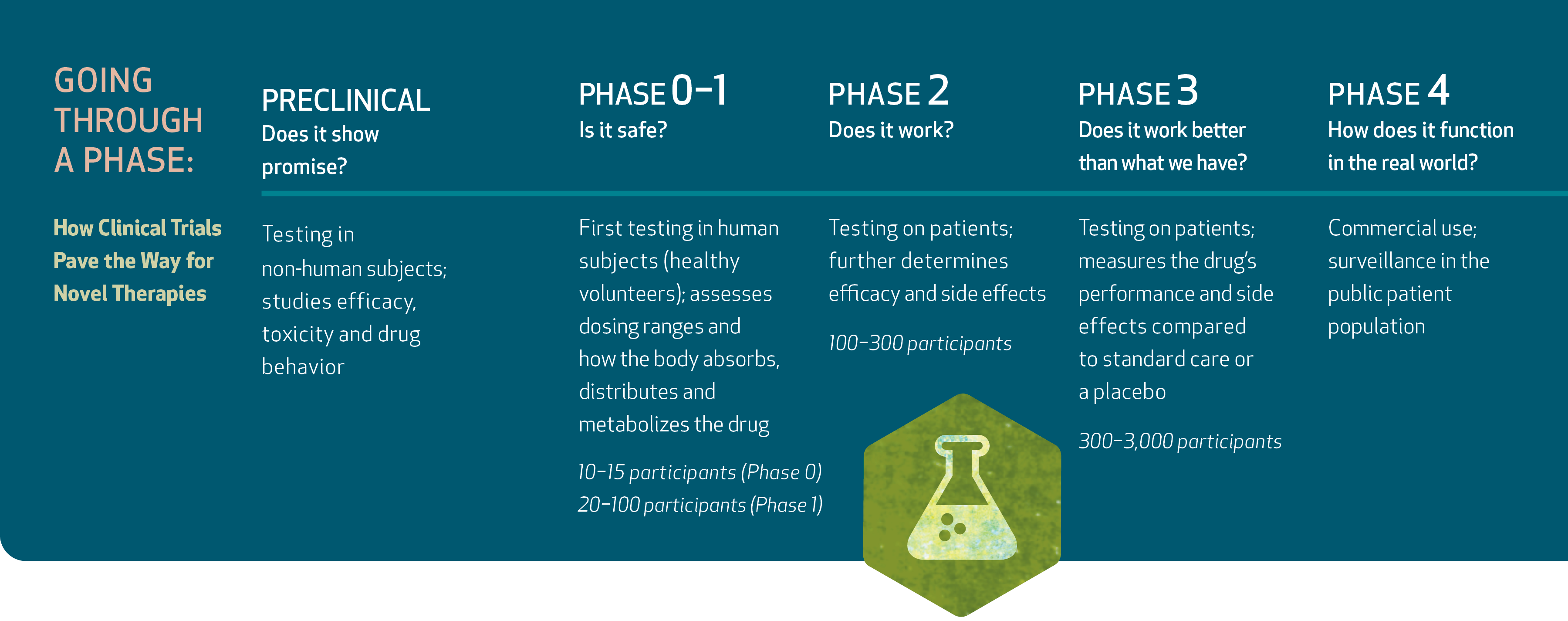
In the world of biomedical research, clinical trials are the gauntlet through which every future cure must march. For promising new therapies, phase 1 trials are a proving ground. These early studies are often a drug’s first-in-human use and begin to answer the many questions that determine whether the agent advances to clinical adoption. Is it safe? Are side effects tolerable? What is the appropriate dose?
Patient populations and research needs are growing at major academic medical centers. Phase 1 and later-stage trials offer access to treatments not yet available elsewhere — and hope when the current standard of care falls short. According to the National Center for Biotechnology Information, the number of registered clinical trials in the U.S. has soared over the last 20 years, today standing at more than 160,000, with nearly 20,000 actively recruiting.
AN INSTRUMENT OF CHANGE
Since 2010, the Cockrell Center for Advanced Therapeutics (CCAT) in the Houston Methodist Research Institute has served as a gateway to crucial clinical testing. Through the long-running generosity of The Cockrell Foundation, CCAT supports all phases of clinical research operations at Houston Methodist and became home to a Clinical Research Phase 1 Unit in 2015. This onsite, early-phase and proof-of-concept trials unit includes exam rooms, private infusion rooms and a specimen-processing lab, all adjacent to inpatient units and a dedicated radiology facility.
“CCAT is really the nexus of everything that a translational research center like Houston Methodist is built around,” explains Edward Jones, president and CEO of the Houston Methodist Research Institute. “It serves as the conduit between the innovation happening in our discovery laboratories and its application at the patient’s bedside.”
Since CCAT’s inaugural trial, an international effort targeting chronic lymphocytic leukemia, the Phase 1 Unit has fueled 205 open clinical trials. Additional Cockrell pilot funding has nurtured the emergent work of talented physician-scientists, enhanced by affiliations with leading academic entities, such as The University of Texas MD Anderson Cancer Center. Meanwhile, new developments in the field of transplant oncology have propelled CCAT to national prominence.
“We see the center as illustrating what can be achieved when you bring brilliant minds together with the resources they need to translate their ideas into lifesaving therapies,” says Janet Cockrell, a director on The Cockrell Foundation board.
STRENGTHENING THE HUMAN ELEMENT
When The Cockrell Foundation made another transformational investment in CCAT in 2025, its board worked closely with Houston Methodist leadership to thoughtfully direct the gift. “We realized that to elevate the potential of the current program, it’s necessary to build out a different type of infrastructure: people,” notes Stephanie Cockrell, executive director of The Cockrell Foundation. “The human element is critically important alongside the physical resources and partnerships already in place.”
Indeed, while Houston Methodist employs nearly 1,500 physicians systemwide, only those who undergo specialized, mentored training on regulatory requirements, research protocol and trial supervision become the credentialed principal investigators who conduct human interventional research. Similarly, clinical staff — including physician assistants and nurse practitioners (collectively termed advanced practice providers, or APPs) — must be rigorously trained on current regulations, technology and best practices.
“This investment in our CCAT investigators and their vital support network radically advances CCAT’s role as a valued research partner across the Texas Medical Center,” says Pauline Todd, director of research for Houston Methodist’s Academic Office of Clinical Trials and CCAT. “And with more than 50% of our patients seen in outlying areas of Houston, it increases synergies across our campuses to expand clinical trial availability.”

INVESTIGATORS IN TRAINING
The new Virginia and Ernest Cockrell Jr. Advanced Therapeutics Development Endowment will launch two highly anticipated training programs. First, the Cockrell Clinical Trialist Development Program will offer much-needed support for advanced independent investigator education for MD and MD-PhD providers with an interest in early-phase clinical studies.
Second, the Cockrell Advanced Practice Provider and PharmD Development Program will equip APP and PharmD sub-investigators to conduct essential patient management, study-related visits and follow-up services. Awardees from partnering departments will receive up to two years of training in clinical trial management in alliance with a mentoring scientist as co-investigator. These advanced clinical partners will serve as an “extension” of busy principal investigators, increasing process efficiencies and lowering overall trial costs.
“One of the things that makes CCAT so special is that it’s not wedded to researchers in just one discipline,” says Jones. “Here, scientists across departments, including cancer, the neurosciences, transplant and orthopedics, interact to share knowledge that serves to inform the others.”
A WORLD-CLASS RESEARCH HUB
As this human infrastructure grows, newly skilled trialists will continue to tap into another integral asset: CCAT’s far-sighted collaboration with the Houston Methodist Ann Kimball & John W. Johnson Center for Cellular Therapeutics. An onsite cellular manufacturing facility, the center supports preparation of commercial cell therapy products — including CAR T cells for infusion at CCAT — dramatically reducing delays associated with contracting external cellular manufacturers.
“Our clinician-researchers have the distinct advantage of rapid translation of new ideas,” observes Todd. “This means that our CCAT trialists can lean ever more into earlier disease intervention and prevention rather than only targeting advanced disease. This has real implications for autoimmune conditions, neurodegenerative disease, diabetes and other domains.”
Sixteen years into CCAT’s mission, Jones reflects on its enduring blueprint as the core of a world-class research hub. “It’s hard to quantify the impact that the Cockrell family, both through their generosity and leadership, has had from one generation to the next at Houston Methodist,” he concludes. “They’ve left an indelible mark on us and, in turn, on thousands of patients in need.”
Discover More








